Life Science and Technology News
Scientists synthesize novel artificial molecules that mimic a cell membrane protein
Scientists at Tokyo Institute of Technology (Tokyo Tech) recently developed an artificial transmembrane ligand-gated channel that can mimic the biological structure and function of its natural counterpart. The findings address one of the roadblocks in biomimetic material research, as the artificial molecule locates in the membrane of a living cell on its own and successfully transports the ions in a regulated manner. This could advance the research in the field of sensing and separation device development.
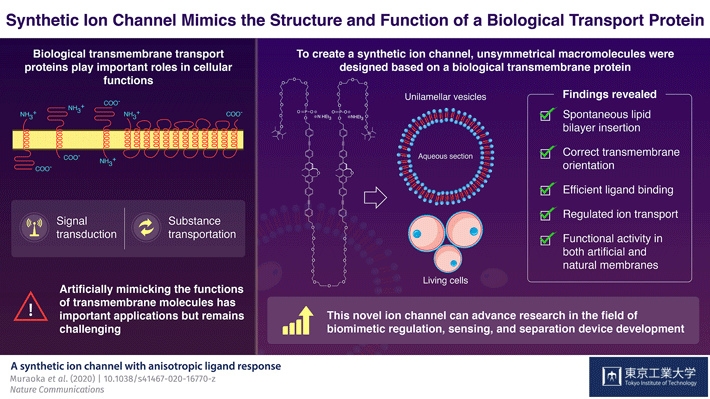
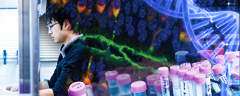
Figure 1. Schematic illustration of the synthetic ion channel which mimics the structure and function of a biological transport protein
In higher organisms, cells and organelles are surrounded by a membrane, which plays a crucial role in not just creating a barrier from the external environment but also mediating exchange of fluids, electrolytes, proteins, and other useful material. Usually, these membranes are composed of water-repelling layers formed by lipid molecules, with various "transmembrane" proteins embedded in this double-layered sheet. These proteins are assembled in a way such that they create unique "gates" or "channels" that open and close in response to selective molecules or ions under specific conditions. These properties of "selectivity" and "sensing capacity" of a biological membrane come from its sophisticated structure, and together they make these membranes an attractive model for the synthesis of novel materials used to develop advanced sensing and separation devices. However, artificially developing such molecular assemblies―that can assemble itself in a membrane in a functionally active orientation―has remained challenging until now.
Advancing the research on artificial molecules, in a study published in Nature Communications![]() , scientists from Tokyo Tech developed a synthetic channel that can mimic the ion-transporting activity of natural ion channels. Prof Kazushi Kinbara and Prof Takahiro Muraoka, the co-authors of the study, explain, "A major obstacle that limits the application of artificial transmembrane molecules is achieving the functionally active orientation. We tried to create a transmembrane molecule that would overcome this difficulty."
, scientists from Tokyo Tech developed a synthetic channel that can mimic the ion-transporting activity of natural ion channels. Prof Kazushi Kinbara and Prof Takahiro Muraoka, the co-authors of the study, explain, "A major obstacle that limits the application of artificial transmembrane molecules is achieving the functionally active orientation. We tried to create a transmembrane molecule that would overcome this difficulty."
To achieve this goal, the scientists focused on the structure of a biological ion channel that spans the membrane multiple times, and used it as the basis to design two artificial molecules. These molecules were composed of both water-repelling structural blocks, called BPO unit, and water-soluble parts called oligoethylene glycol chains. These structural features lend these artificial molecules the capacity to self-aggregate when embedded in membranes. The molecules also contained phosphate groups that further helped them to achieve the correct orientation across the membranes.
Next, the scientists focused on one of the two molecules, to analyze its structural properties. They observed that when suitable bait-like "ligand" molecules were added to a solution containing the artificial molecule, they successfully bound to the structure―confirming that the structure was indeed functionally active. Moreover, when these molecules were introduced to a preformed membrane, they could insert and orient themselves in the membrane on their own. In the presence of the specific ligands, the membrane-embedded macromolecules changed their structures and transported ions, including lithium, potassium, and sodium ions. Because the synthetic molecule showed promising results with artificial membranes, the scientists then tested it in living cells. Using a technique called fluorescence microscopy, they observed that the macromolecule showed the same functional properties, including differential ligand binding and regulated ion transport activities, in biological membranes too!
Taken together, the study shows how an artificially designed molecule can self-assemble, localize, orient, and mimic the biological ion transport process. These findings can potentially spur advances in the field of biomimetic regulation. The authors optimistically conclude, "The promising results of our study addressed a persistent limitation that blocked the way of using artificial biomimetic membrane proteins in applied fields."
- Reference
| Authors |
Takahiro Muraoka1,2, Daiki Noguchi3, Rinshi S. Kasai4, Kohei Sato1, Ryo Sasaki1, Kazuhito V. Tabata5, Toru Ekimoto6, Mitsunori Ikeguchi 6,7, Kiyoto Kamagata3, Norihisa Hoshino3, Hiroyuki Noji5, Tomoyuki Akutagawa3, Kazuaki Ichimura1 and Kazushi Kinbara1,3 * Corresponding author's email: Takahiro Muraoka: tmuraoka@bio.titech.ac.jp / Kazushi Kinbara: kkinbara@bio.titech.ac.jp |
|---|---|
| Title of original paper | A synthetic ion channel with anisotropic ligand response |
| Journal | Nature Communications |
| DOI | 10.1038/s41467-020-16770-z |
| Affiliations |
1 School of Life Science and Technology, Tokyo Institute of Technology 2 Precursory Research for Embryonic Science and Technology, Japan Science and Technology Agency 3 Institute of Multidisciplinary Research for Advanced Materials, Tohoku University 4 Institute for Frontier Life and Medical Sciences, Kyoto University 5 Department of Applied Chemistry, School of Engineering, The University of Tokyo 6 Graduate School of Medical Life Science, Yokohama City University 7 Medical Sciences Innovation Hub Program RIKEN |
- Kinbara Research Group
- Researcher Profile | Tokyo Tech STAR Search - Kazushi Kinbara
- Department of Life Science and Technology, School of Life Science and Technology
- Latest Research News
School of Life Science and Technology
—Unravel the Complex and Diverse Phenomena of Life—
Information on School of Life Science and Technology inaugurated in April 2016
Further Information
Professor Kazushi Kinbara
Department of Life Science and Technology,
School of Life Science and Technology,
Tokyo Institute of Technology
E-mail : kinbara.k.aa@m.titech.ac.jp
Tel +81-45-924-5781
Contact
Public Relations Group, Tokyo Institute of Technology
E-mail : media@jim.titech.ac.jp
Tel +81-3-5734-2975