Life Science and Technology News
Boron carrier for targeted tumour therapy
Scientists at Tokyo Institute of Technology have developed a boron carrier for use in targeted radiation treatment for cancerous tumours. The carrier is based on a common blood plasma protein, meaning it can be tailored to individual patients thus lessening the chances of blood contamination.
Targeted radiation-based therapies for treating cancerous tumours such as 'boron neutron capture therapy' (BNCT), rely on the efficient and effective delivery of the capture agent (in this case, boron) to the tumour. The agent must collect in the tumour in high enough concentrations to trigger an effective reaction during thermal neutron irradiation.
Delivering the agent to tumours safely, while limiting the chances of it being distributed in the bloodstream and major organs, is a major challenge. Hiroyuki Nakamura, who teaches Human Centered Science and Biomedical Engineering, and co-workers at the Tokyo Institute of Technology, in collaboration with researchers across Japan, decided to test the ability of a common blood plasma protein known as albumin to carry boron to tumours during BNCT.
Albumin flows readily to and accumulates in malignant tissues; in fact, it is a major source of nutrition for growing tumours. Tumours therefore attract albumin, making the protein an ideal candidate for carrying drugs — it has already been used as a drug carrier for breast, lung and pancreatic cancers. However, its ability to transport boron for BNCT remains untested.
Nakamura's team developed a tiny capsule, made from maleimide-functionalized closo-dodecaborate (MID), and trialled its ability to bind with albumin and to carry boron. They found that MID successfully binds with both cysteine and lysine residues in albumin, creating a stable carrier for boron that the team called MID-AC.
They tested MID-AC on 26 tumour-bearing mice. The team found that the boron concentrated highly and efficiently in tumours; the active uptake of boron by the albumin led to double the concentration of boron reaching tumours than previous systems. Levels of boron in the bloodstream and organs were very low, indicating a viable targeted delivery system. Thermal irradiation was then carried out on the mice, and led to the significant suppression of tumour growth even at low boron levels.
This technique could limit existing problems of using blood-based products in cancer treatment. Albumin could be taken from patients in hospital prior to treatment, then used in MID-AC to carry boron to tumours in the patients' own bodies. This will limit contamination and encourage the acceptance of the system by the patients' immune systems.
Background
Boron neutron capture therapy
Boron neutron capture therapy (BNCT) is a non-invasive method for the targeted destruction of cancer cells in tumours. The two-step technique firstly relies on the effective delivery of boron — a non-radioactive isotope capable of capturing neutrons to trigger a nuclear reaction — to tumours. The patient is injected with boron carriers which make their way through the bloodstream to the tumour. Creating carriers that target tumours specifically without discharging boron in healthy tissues has been a significant challenge for scientists. Once the boron has accumulated in sufficient levels in the tumour, the patient undergoes thermal neutron irradiation — the resulting miniature nuclear reaction inside the tumour blasts the cancer cells and destroys them.
So far, two small boron molecules, such as mercaptoundecahydrododecaborate (BSH) and L-p-boronophenylalanine (L-BPA), have been used as boron carriers for clinical treatment of brain tumors, melanoma, head and neck cancers, and other cancers in BNCT.
Implications of the current study
The MID-AC system is based on a common blood protein called albumin, which is found in abundant levels in blood plasma. Nakamura's team developed a boron carrier which uses albumin to transport the agent to the tumour. As a result, the system could greatly enhance current BNCT delivery, because if patients' own albumin is used to carry the boron to the tumour, there is a far smaller chance of blood contamination — a risk when using blood products from other people.
Further, the success of the system lies in the fact that it makes use of albumin's natural propensity to gather in tumours. This means that boron levels in surrounding healthy cells and organs is limited, reducing the risk of side effects during radiation-based cancer therapy.
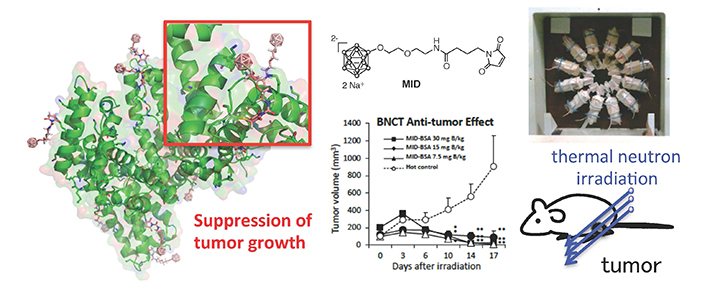
Figure. Efficient BNCT Effects of MID-AC toward Tumor-Bearing Mice
Reference
| Authors | S. Kikuchia, D. Kanohb, S. Satoa, Y. Sakuraic, M. Suzukid, H. Nakamuraa |
|---|---|
| Journal | Journal of Controlled Release, vol 237 (2016) |
| Title of original paper | Maleimide-Functionalized closo-Dodecaborate Albumin Conjugates (MID-AC): The Unique Ligation at both Cysteine and Lysine Residues Enabling to Efficient Boron Delivery to Tumor for Neutron Capture Therapy |
| DOI | 10.1016/j.jconrel.2016.07.017 |
| Affiliations | aLaboratory for Chemistry and Life Science, Institute of Innovative Research, Tokyo Institute of Technology bDepartment of Chemistry, Faculty of Science, Gakushuin University cDepartment of Radiation Life Science and Radiation Medical Science, Kyoto University Research Reactor Institute dParticle Radiation Oncology Research Center, Kyoto University Research Reactor Institute |
- Nakamura-Fuse Group
- Researcher Profile | Tokyo Tech STAR Search - Hiroyuki Nakamura
- Laboratory for Chemistry and Life Science, Institute of Innovative Research
- Kyoto University Research Reactor Institute
- Latest Research News
School of Life Science and Technology
—Unravel the Complex and Diverse Phenomena of Life—
Information on School of Life Science and Technology inaugurated in April 2016
Further information
Professor Hiroyuki Nakamura
Laboratory for Chemistry and Life Science, Institute of Innovative Research
Email hiro@res.titech.ac.jp
Tel +81-45-924-5244