Materials Science and Engineering News
Finding new controlling method for properties of glass
Tiny amount of electron anions reduces the glass transition temperature more than 100 degrees
Electride is a compound in which electrons serve as anions. A group of Tokyo Tech and Pacific Northwest National Laboratory led by Professor Hideo Hosono found that electride glass exhibits a property which has not seen in conventional glasses and succeeded in clarifying the mechanism.
The glass transition temperature is generally determined by the ratio of network forming component to the network breaking component. i.e., the chemical composition of a bulk glass. Researchers successfully prepared the glass of 12CaO・7Al2O3 electride and found that the glass transition temperature is distinctly reduced with increasing the concentration of electron anions. When only 3% of oxygen ion are replaced by electrons, the glass transition temperature was reduced by more than 100 degrees, which is almost 2 times as large as the magnitude of glass transition temperature change when the CaO content is varied from 56% to 70% (the whole glass formation range). Ab initio molecular dynamics simulation reproduced this result and elucidated that electron anions are so mobile than the constituent ions that local temperature around them is rather higher than the bulk.
Several novel properties are expected for electride glasses which is a novel class of amorphous materials along with applications.
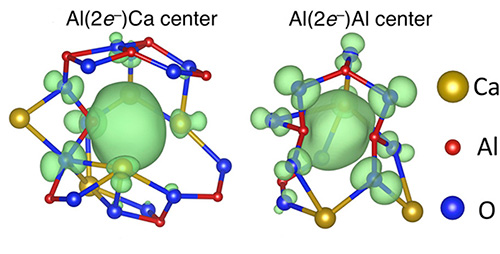
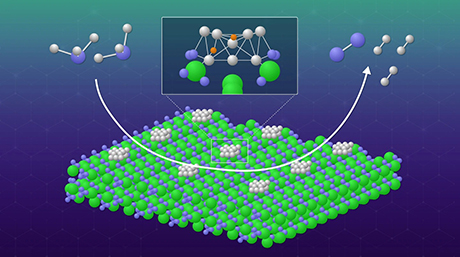
Figure. Electron Anions
Electron anions (center green blob) pair up in the center of molecular cages and lower the temperature at which glass forms in C12A7 electride.
©Courtesy of Pacific Northwest National Laboratory
Program Information
- JST ACCEL
- R&D Project "Materials Science and Application of Electrides"
Reference
| Authors: | Lewis E. Johnson1, Peter V. Sushko1*, Yudai Tomota2 , Hideo Hosono2* *corresponding authors |
|---|---|
| Title of original paper: | Electron anions and the glass transition temperature |
| Journal: | Proceedings of the National Academy of Sciences of the United States of America |
| DOI : | 10.1073/pnas.1606891113 |
| Affiliations : | 1 Physical Sciences Division, Physical & Computational Sciences Directorate, Pacific Northwest National Laboratory 2 Materials Research Center of Element Strategy, Tokyo Institute of Technology |
- FACES: Tokyo Tech Researchers, Issue 16 - Hideo Hosono, Part 1
- FACES: Tokyo Tech Researchers, Issue 16 - Hideo Hosono, Part 2
- Professor Hosono receives 2016 Japan Prize, gives commemorative lecture
- AGC starts mass-production of sputter target of C12A7 electride created by Tokyo Tech
- Hosono-Kamiya Laboratory
- Researcher Profile | Tokyo Tech STAR Search - Hideo Hosono
- Laboratory for Materials and Structures, Institute of Innovative Research
- Institute of Innovative Research
- Press Release "Super cement's secret"
- Pacific Northwest National Laboratory (PNNL)
- Latest Research News
Further information
Professor Hideo Hosono
Institute of Innovative Research, Tokyo Institute of Technology
Email hosono@msl.titech.ac.jp
About Program
Daichi Terashita
ACCEL Group, Department of Innovation Research, JST
Email suishinf@jst.go.jp
Tel +81-3-5734-2975