Materials Science and Engineering News
Reusable Catalyst Makes C–H Bond Oxidation Using Oxygen Easier and More Efficient
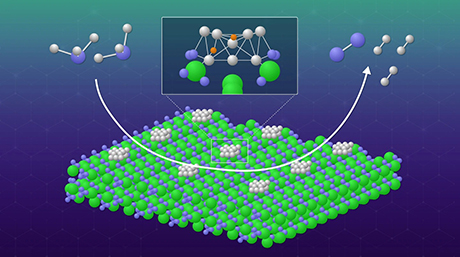
The selective oxidation of C–H bonds using oxygen has become a much simpler and more sustainable endeavor, thanks to a novel manganese (Mn)-based catalyst developed by scientists at Tokyo Institute of Technology (Tokyo Tech). Consisting of reusable Mg6MnO8 nanoparticles with unprecedented surface area, their catalyst enables the desired oxidative reactions to occur at mild temperature and pressure conditions without the need for toxic additives, opening doors to more energy-efficient and environmentally friendly chemistry applications.

In the chemical industry, the selective cleavage and oxidation of carbon–hydrogen (C–H) bonds, called "oxidative C–H functionalization" is an essential step in the production of many solvents, polymers, and surfactants, as well as intermediate compounds for agrochemicals and pharmaceuticals. Ideally, one would want to use oxygen (O2) as the only oxidant in this process to avoid using more expensive and environmentally taxing substances, such as hydrogen peroxide (H2O2), chlorine (Cl2), or nitric acid (HNO3).
However, using O2 as the oxidant entails some unresolved problems. While some progress has been made in the field of recoverable and reusable catalysts, most heterogeneous systems require high reaction temperatures, high O2 pressures, or the use of toxic additives. In turn, this cripples the scope of potential applications, scalability, and efficiency of these catalytic systems.
Against this backdrop, a team of scientists from Tokyo Tech, led by Associate Professor Keigo Kamata, recently found a promising catalyst for oxidative C–H functionalization. As explained in their paper published in ACS Applied Materials & Interfaces![]() , they inferred that isolated manganese (Mn) species fixed in a crystalline matrix could constitute a high-performance heterogeneous catalyst even at mild reaction conditions, based on previous knowledge.
, they inferred that isolated manganese (Mn) species fixed in a crystalline matrix could constitute a high-performance heterogeneous catalyst even at mild reaction conditions, based on previous knowledge.
Accordingly, they investigated the catalyst murdochite-type Mg6MnO8, a rock salt structure of magnesium oxide (MgO) with one eighth of the Mg2+ ions replaced with Mn4+ ions and another eighth replaced with vacancies, resulting in a crystal with Mn ions and vacancies orderly occupying alternating layers. Using a cost-effective sol–gel method aided by malic acid, the team prepared Mg6MnO8 nanoparticles with a very high surface area. Dr. Kamata elaborates: "The specific surface area of our Mg6MnO8 catalyst was 104 m2/g, about seven times higher than that of Mg6MnO8 synthesized using previously reported methods."
The researchers also demonstrated, through numerous experiments, that their Mg6MnO8 nanoparticles could efficiently catalyze the selective oxidation of C–H of various alkylarene compounds even under mild reaction conditions, namely 40℃ and atmospheric pressure. The yield of the final products was also higher than that obtained using existing Mn-based catalysts. To top things off, the Mg6MnO8 nanoparticles could be easily recovered via filtration and then reused without any apparent loss in catalytic activity after multiple cycles.
Finally, the team sought to understand why their proposed catalyst performed so well through a series of kinetic and mechanistic studies. They concluded that the isolation of redox sites (Mn species, in this case) in a crystalline base matrix (MgO) was a particularly important feature to achieve oxidative C–H functionalization using O2 at mild conditions.
Satisfied with the results and their findings, Dr. Kamata speculates: "Our approach constitutes a promising strategy for the development of highly efficient heterogeneous oxidation systems with wide substrate scopes."
Let us hope this study paves the way to more efficient and environment-friendly catalysts for organic chemistry applications.
- Reference
| Authors : | Eri Hayashi, Takatoshi Tamura, Takeshi Aihara, Keigo Kamata*, and Michikazu Hara* |
|---|---|
| Title of original paper : | Base-Assisted Aerobic C–H Oxidation of Alkylarenes with a Murdochite-Type Oxide Mg6MnO8 Nanoparticle Catalyst |
| Journal : | ACS Applied Materials & Interfaces |
| DOI : | 10.1021/acsami.1c20080 |
| Affiliations : | Laboratory for Materials and Structures, Institute of Innovative Research, Tokyo Institute of Technology |
* Corresponding authors' emails: kamata.k.ac@m.titech.ac.jp ; hara.m.ae@m.titech.ac.jp
- Dethroning Electrocatalysts for Hydrogen Production with Inexpensive Alternative Material | Tokyo Tech News
- An enhanced ruthenium-based catalyst for primary amine synthesis | Tokyo Tech News
- Way, Shape and Form: Synthesis Conditions Define the Nanostructure of Manganese Dioxide | Tokyo Tech News
- Fuelling the World Sustainably: Synthesizing Ammonia using Less Energy | Tokyo Tech News
- Green catalysts with Earth-abundant metals accelerate production of bio-based plastic | Tokyo Tech News
- Barium ruthenate: A high-yield, easy-to-handle perovskite catalyst for the oxidation of sulfides | Tokyo Tech News
- A ruthenium-based catalyst with highly active, flat surfaces outperforms metal-based competitors | Tokyo Tech News
- Reusable ruthenium-based catalyst could be a game-changer for the biomass industry | Tokyo Tech News
- Chemoselective acetalization by a bifuncional cerium phosphate catalyst | Tokyo Tech News
- Development of highly active and stable ammonia synthesis catalyst under low temperatures | Tokyo Tech News
- Hara & Kamata Laboratory
- Keigo Kamata | Researcher Finder - Tokyo Tech STAR Search
- Michikazu Hara | Researcher Finder - Tokyo Tech STAR Search
- Laboratory for Materials and Structures
- Institute of Innovative Research (IIR)
- Materials Science and Engineering Undergraduate Major|Education|Department of Materials Science and Engineering, School of Materials and Chemical Technology
- Materials Science and Engineering Graduate Major|Education|Department of Materials Science and Engineering, School of Materials and Chemical Technology
- Energy Science and Engineering Graduate Major|Education|Department of Materials Science and Engineering, School of Materials and Chemical Technology
- Latest Research News
Further Information
Associate Professor Keigo Kamata
Institute of Innovative Research, Tokyo Institute of Technology