Life Science and Technology News
【Labs spotlight】 Yuasa Laboratory
Sugar-, nucleic acid-, & photo-based biomolecules
The Department has a variety of laboratories for Life Science and Technology, in which cutting-edge innovative research is being undertaken not only in basic science and engineering but also in the areas of medicine, pharmacy, agriculture, and multidisciplinary sciences.
This "Spotlight" series features a laboratory from the Department and introduces you to the laboratory's research projects and outcomes. This time we focus on Yuasa Laboratory.

Areas of Supervision
Primary/Life Science and Technology
Secondary/Science and Technology for health Care and Medicine, Human Centered Science and Biomedical Engineering
Professor Hideya Yuasa![]()
| Degree | PhD 1991, Tokyo Institute of Technology |
|---|---|
| Areas of Research | Bioorganic Chemistry, Photochemistry. |
| Keywords | Carbohydrate, Lanthanide, Phosphorescence, Molecular Devices. |
| Website | Yuasa and Ohkubo Laboratory (Japanese) |
Research interest
1. Photodynamic therapy of cancer using near-infrared and lanthanide nanoparticles.
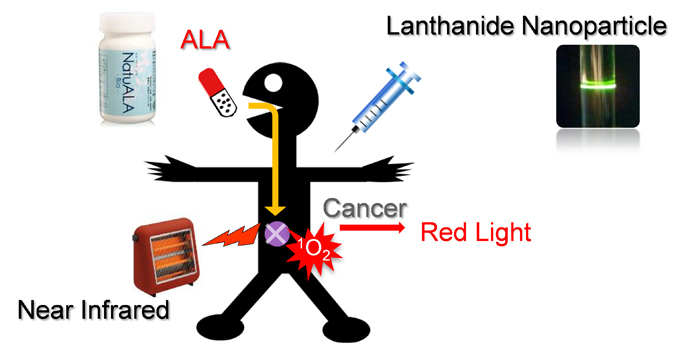
A. Shimoyama, et al., Access to a Novel Near-Infrared Photodynamic Therapy by the Combined Use of 5-Aminolevulinic Acid and Lanthanide Nanoparticles, Photodiagnosis and Photodynamic Therapy, 10, 607-614 (2013).
2. Stimuli-responsive liposome using a motional molecular device.

J. Takeuchi, et al., A Ring-Flippable Sugar as a Stimuli-Responsive Component of Liposomes, Chem. Asian J. in press (2015).
3. New phosphorescence mechanism.
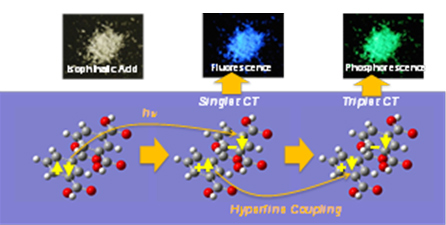
S. Kuno, et al., Visible Room-Temperature Phosphorescence of Pure Organic Crystals via a Radical-Ion-Pair Mechanism, submitted.
Research findings
Selected publications
- [1] S. Kuno, T. Kanamori, Z. Yijing, H. Ohtani and H. Yuasa. Long Persistent Phosphorescence of Crystalline Phenylboronic Acid Derivatives: Photophysics and a Mechanistic Study. ChemPhotoChem, 1, 102-106 (2017).
- [2] T. Kanamori, T. Sawamura, T. Tanaka, I. Sotokawa, R. Mori, K. Inada, A. Ohkubo, S. I. Ogura, Y. Murayama, E. Otsuji and H. Yuasa. Coating lanthanide nanoparticles with carbohydrate ligands elicits affinity for HeLa and RAW264.7 cells, enhancing their photodamaging effect. Bioorg. Med. Chem., 25, 743-749 (2017).
- [3] J. Takeuchi, A. Ohkubo and H. Yuasa. A ring-flippable sugar as a stimuli-responsive component of liposomes. Chem. Asian. J., 10, 586-594 (2015).
- [4] T. Sawamura, T. Tanaka, H. Ishige, M. Iizuka, Y. Murayama, E. Otsuji, A. Ohkubo, S. Ogura and H. Yuasa. The Effect of Coatings on the Affinity of Lanthanide Nanoparticles to MKN45 and HeLa Cancer Cells and Improvement in Photodynamic Therapy Efficiency. Int. J. Mol. Sci., 16, 22415-22424 (2015).
- [5] A. Ohkubo, K. Yamada, Y. Ito, K. Yoshimura, K. Miyauchi, T. Kanamori, Y. Masaki, K. Seio, H. Yuasa and M. Sekine. Synthesis and triplex-forming properties of oligonucleotides capable of recognizing corresponding DNA duplexes containing four base pairs. Nucleic Acids Res., 43, 5675-5686 (2015).
- [6] S. Kuno, H. Akeno, H. Ohtani and H. Yuasa. Visible room-temperature phosphorescence of pure organic crystals via a radical-ion-pair mechanism. Phys. Chem. Chem. Phys., 17, 15989-15995 (2015).
- [7] S. Hososhima, H. Yuasa, T. Ishizuka, M. R. Hoque, T. Yamashita, A. Yamanaka, E. Sugano, H. Tomita and H. Yawo. Near-infrared (NIR) up-conversion optogenetics. Sci. Rep., 5, 16533 (2015).
- [8] A. Shimoyama, H. Watase, Y. Liu, S. Ogura, Y. Hagiya, K. Takahashi, K. Inoue, T. Tanaka, Y. Murayama, E. Otsuji, A. Ohkubo and H. Yuasa. Access to a novel near-infrared photodynamic therapy through the combined use of 5-aminolevulinic acid and lanthanide nanoparticles. Photodiagnosis Photodyn. Ther., 10, 607-614 (2013).
- [9] Y. Liu, T. Kobayashi, M. Iizuka, T. Tanaka, I. Sotokawa, A. Shimoyama, Y. Murayama, E. Otsuji, S. Ogura and H. Yuasa. Sugar-attached upconversion lanthanide nanoparticles: a novel tool for high-throughput lectin assay. Bioorg. Med. Chem., 21, 2832-2842 (2013).
- [10] Y. Yuzawa, H. Nishihara, T. Haraguchi, S. Masuda, M. Shimojima, A. Shimoyama, H. Yuasa, N. Okada and H. Ohta. Phylogeny of galactolipid synthase homologs together with their enzymatic analyses revealed a possible origin and divergence time for photosynthetic membrane biogenesis. DNA Res., 19, 91-102 (2012).
- [11] H. Tsutsumi, H. Ohkusa, H. Park, T. Takahashi, H. Yuasa and H. Mihara. Gold nanoparticles conjugated with monosaccharide-modified peptide for lectin detection. Bioorg. Med. Chem. Lett., 22, 6825-6827 (2012).
- [12] L. Y. Yang, Y. Kawada, L. Bai, D. Kubota and H. Yuasa. 2-Oxabutane as a substitute for internal monomer units of oligosaccharides to create lectin ligands. Org. Biomol. Chem., 9, 6579-6586 (2011).
- [13] S. Masuda, J. Harada, M. Yokono, Y. Yuzawa, M. Shimojima, K. Murofushi, H. Tanaka, H. Masuda, M. Murakawa, T. Haraguchi, M. Kondo, M. Nishimura, H. Yuasa, M. Noguchi, H. Oh-Oka, A. Tanaka, H. Tamiaki and H. Ohta. A monogalactosyldiacylglycerol synthase found in the green sulfur bacterium Chlorobaculum tepidum reveals important roles for galactolipids in photosynthesis. Plant Cell., 23, 2644-2658 (2011).
- [14] L. Y. Yang, T. Haraguchi, T. Inazawa, S. Kajiwara and H. Yuasa. Synthesis of a novel class of glycocluster with a cyclic α-(1→6)-octaglucoside as a scaffold and their binding abilities to concanavalin A. Carbohydr. Res., 345, 2124-2132 (2010).
- [15] H. Yuasa, M. Izumi and H. Hashimoto. Thiasugars: potential glycosidase inhibitors. Curr. Top. Med. Chem., 9, 76-86 (2009).
- [16] H. Yuasa, T. Haraguchi and T. Itagaki. A Facile Latex Agglutination Lectin Assay (LALA) for Weakly Binding Ligands. J. Carbohydr. Chem., 28, 78-93 (2009).
- [17] N. Mitsuhashi and H. Yuasa. A Novel Galactosyltransferase Inhibitor with Diamino Sugar as a Pyrophosphate Mimic. Eur. J. Org. Chem., 2009, 1598-1605 (2009).
- [18] M. Izumi, H. Yuasa and H. Hashimoto. Bisubstrate analogues as glycosyltransferase inhibitors. Curr. Top. Med. Chem., 9, 87-105 (2009).
- [19] H. Yuasa, O. Tsuruta, T. Izumi, T. Takahara, M. Izumi and H. Hashimoto. β-Selective Glycosidation of a 5-Thioglucosamine Derivative. Chem. Lett., 37, 1288-1289 (2008).
- [20] H. Yuasa, Y. Tanetani, T. Miyazaki, M. Izumi and H. Hashimoto. Syntheses and Glycosidation Reactions of 6-Thiosialosyl Donors. Lett. Org. Chem., 5, 262-264 (2008).
- [21] H. Yuasa, Y. Suga and H. Hashimoto. Glycosidation Reactions of 5-Thioxylopyranosyl Donors. Lett. Org. Chem., 5, 429-431 (2008).
- [22] H. Yuasa, M. Jouyabu, N. Mitsuhashi and H. Hashimoto. Syntheses of 5-Thio-D-Mannose from Petrochemicals and a Disaccharide Analog Containing It. Res. Lett. Org. Chem., 2008, 1-4 (2008).
- [23] T. Miyazaki, D. Hiyoshi, M. Izumi, H. Hashimoto and H. Yuasa. Stereoselective Synthesis of 5-Thio-β-L-Fucopyranosyl Phosphate. Lett. Org. Chem., 5, 576-578 (2008).
- [24] H. Yuasa, O. Tsuruta and H. Hashimoto. A novel 5-thioglycosylation method with 1,5-dithioglycosyl donors: relevance to exo- versus endocyclic activation. Tetrahedron Lett., 48, 7953-7956 (2007).
- [25] H. Yuasa, H. Honma, H. Hashimoto, M. Tsunooka and K. Kojima-Aikawa. Pentamer is the minimum structure for oligomannosylpeptoids to bind to concanavalin A. Bioorg. Med. Chem. Lett., 17, 5274-5278 (2007).
- [26] H. Yuasa, N. Fujii and S. Yamazaki. A novel proton-selective sensor based on a sugar with hinge flexibility. Org. Biomol. Chem., 5, 2920-2924 (2007).
Contact
Professor Hideya Yuasa
Room 803, J2 building, Suzukakedai campus
E-mail : hyuasa@bio.titech.ac.jp
*Find more about the lab and the latest activities at the lab site (Japanese)![]() .
.
*May 1, 2025:Some of the content has been updated with the latest information.